RNA - Peptide Interactions
Supramolecular RNA-peptide chemistry: We are investgating the supramolecular interactions of RNA with peptides. This will deepen our understanding of how proteins bind to RNA which is has a number of implications in biology and medicine ranging from gene regulation and CRISPR systems to understanding mRNA trafficing in the body.
RNA-peptide condensates / aggregates: It is now clear that the role of RNA in biology goes way beyond of that of simply being a "messenger" or indeed how genetic information is transcripted to proteins. So-called non-coding RNA form various complex gel-like aggregates (condensates) with a range of proteins within the cell. The condensates have which in turn appear to have a range of regularitory, processing and protective functions within the cell and misregulation of these condensates could be the ultimate cause of complex diseases ranging from various forms of cancer to neurodegenerative diseases including Alzheimer's.
Building on our work on gels formed by peptide, our research in this area is therefore aimed at unravling at the molecular level how various forms of RNA interact with peptides and proteins.
Project example: The role of aromatic interactions in RNA-peptide aggregations
In collaboration with Dr Anna Wang and Dr Albert Fahrebach and A/Prof. Christopher Marquis (BABS UNSW)
Peptides and Nanomedicine
A large part of the research in the Thordarson group revolves around peptides and how they can be utilised on their own as well as in large supramolecular structures. Our group focuses on peptide-based hydrogels and how the manipulation of these hydrogel properties can result in materials ideal for a wide range of applications such as catalysis and 3D cell culture. Our group also utilises peptides as small molecules for drug delivery and making unique photo-switches."
Project example: Light-switchable peptide materials for medical applications
In collaboration with A/Prof. Jonathon Beves and A/Prof. Kris Kilian
Project example: Peptide-functionalised nanoparticles for targeted delivery of cancer drugs and siRNA
In collaboration with Prof. Maria Kavallaris (Children's Cancer Institute) and A/Prof. Joshua McCarroll (Children's Cancer Institute)/
Origin of Life and Systems Chemistry
Exploring the role of self-assembly in how life originated and how we can make life-like systems
Project example: Do Liposomes enhance the catalytic efficiency of RNA and peptides?
In collaboration with Dr Albert Fahrenbach and Prof. Martin Van Kranendonk (BEES, UNSW)
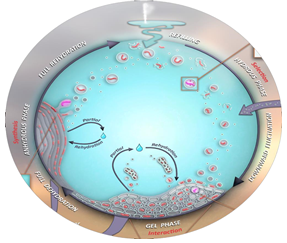
Recently, Origin of Life research2 has started to turn its attention to a new hypothesis for how complexity could have arisen from a “pre-biotic soup” of chemicals. Dubbed here the “geyser” model, it focuses on how hydration/dehydration (HD) cycles in a geothermal pond (fed by geyser activity), could generate liposomes that encapsulate peptides and/or RNA of increasing complexity with each HD cycle (See also the Front Cover article of the Scientific American in August 2017).3 One of the many questions that this model begs, is if the liposomal environment might enhance the catalytic activity of peptides and RNA. In this project, you investigate whether encapsulating catalytically active RNA or peptides in liposomes changes their catalytic activity. This project will not only give you insight into what is arguably one of the most important questions in science, i.e., how did life originate, but you will also gain valuable experience in synthesis, self-assembly and the chemistry of peptide and RNA biomolecules.
References
- Pall Thordarson, “Emergence of Life” in Encyclopedia of Supramolecular Chemistry: eds: Jerry L. Atwood, Jonathan W. Steed, Marcel Dekker Inc., New York, 2004, 528-534.
- Martin Van Kranendonk, David W. Deamer and Tara Djokic, Life Springs, Scientific American, August 2017, 28-35.
Complexity in Supramolecular Chemistry
Investigating the fundamental aspects of host-guest interactions
Open Science / Data Analysis
Developing FREE online tools for data analysis and storage.
Promoting open science and good data analysis practices
The OpenDataFit project including the popular supramolecular.org website